Apollonian
Guest Columnist
Here's fm the Ajax Jewsn (Infowars.com) contest for best satirical efforts on covid madness ginned-up by globalists, big-Pharma, and Jews-media

Here is Sky News report.I found out that his [Fauci’s] agencies had funded 60 projects at the Wuhan Institute of Virology.
I found out that he wrote a paper back in 2012 where he argued that gain of function research that’s genetically manipulating coronaviruses to make them even more dangerous and more transmissable, Fauci said that this was worth the risk of a pandemic.
And he even funded research, his agency funded coronavirus research at the Wuhan Institute of Virology, in conjunction with the Chinese military.

New documents show that just 18 months before the first Covid-19 cases appeared, researchers had submitted plans to release skin-penetrating nanoparticles containing “novel chimeric spike proteins” of bat coronaviruses into cave bats in Yunnan, China.
They also planned to create chimeric viruses, genetically enhanced to infect humans more easily, and requested $14million from the Defense Advanced Research Projects Agency (Darpa) to fund the work.

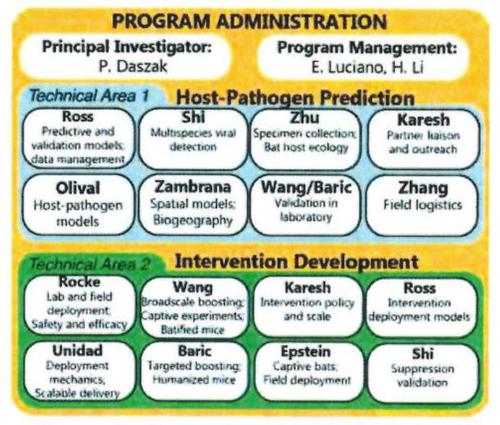
Grant documents show that the team also had some concerns about the vaccine programme and said they would “conduct educational outreach … so that there is a public understanding of what we are doing and why we are doing it, particularly because of the practice of bat-consumption in the region”.
Angus Dalgleish, Professor of Oncology at St Georges, University of London, who struggled to get work published showing that the Wuhan Institute of Virology (WIV) had been carrying out “gain of function” work for years before the pandemic, said the research may have gone ahead even without the funding.
“This is clearly a gain of function, engineering the cleavage site and polishing the new viruses to enhance human cell infectibility in more than one cell line,” he said. -Telegraph
Previous articles provided supporting discussion for the claim that the aforementioned segment may have been a subtle attempt for public health officials and/or politicians to legally cover their plans to “convene” a national, multi-year pandemic exercise.Convene a Unified, National Response and Recovery to Public Health Emergencies and Disasters
ASPR, in coordination with HHS and Federal partners, leads the nation’s public health and medical preparedness and response and its health and social services recovery efforts, as delegated by Emergency Support Function #8, and the Health and Social Services Recovery Support Function. These efforts provide unified national leadership and guidance to public health and health care stakeholders before, during, and after disasters. We recognize that SLTT stakeholders are on the front line during response and may need timely and appropriate Federal support to augment their resources and capacity. Therefore, it is critical to improve situational awareness and coordination across the Federal interagency and with regional and SLTT partners.
In addition, we will continue work with international partners to make sure our preparedness and response efforts are aligned with those of the global community. We will sustain efforts to organize, train, equip, and exercise response capabilities. We will identify health care readiness standards to assess coalition readiness status and ensure exercises test system resilience against routine and catastrophic threats. (Page 10; emphasis added)
In other words, the ASPR’s Strategic Plan appears to be stating that it is a type of legal document supporting the “implementation” of other pandemic laws and legal documents. This is significant because it is more of a “paper trail,” so to speak, of legal actions or documents leading up to and during COVID-19.is aligned to and is designed to support the implementation of a number of different higher-order statutes, executive orders, policies, strategies, and plans, including, but not limited to: the National Security Strategy (NSS), the National Health Security Strategy 2019-2022 (NHSS), the National Biodefense Strategy (NBS), the HHS Strategic Plan, FY2018-2022, and the Pandemic and All-Hazards Preparedness and Advancing Innovation Act (PAHPAIA) of 2019. (Page 2)
Within the same objective of strengthening “response and recovery operations,” the document states thatObjective 2.3: Strengthen Response and Recovery Operations
ASPR continually strives to improve delivery of public health and medical assistance to communities impacted by disaster throughout the mission continuum. Successful preparedness for, response to, and recovery from PHEs [Public Health Emergencies] and other incidents requires accurately identifying gaps, and subsequent planning and preparedness activities to address them. (Page 30, emphasis added)
Now, compare the emphasized segments from the ASPR Strategic Plan for 2020-2023 with the National Health Security Strategy 2019-2022, beginning with a sub-objective and abridged as follows:ASPR will pursue the following strategies to strengthen its disaster response capabilities:
Fully implement, test, and validate the various components of the ASPR Incident Response Framework, including development, implementation and ongoing exercising of all functional annexes (Information Management, Planning, Resource Coordination, and Finance & Administration) and other supporting annexes, operational doctrine, and SOPs [standard operating procedures]
[…]
Establish a financially sustainable, multi-year and multi-level exercise program designed to test policies, plans, and capabilities and achieve and sustain national preparedness to prevent, respond to, and recover from all-hazards disasters (Page 31, emphasis and hyperlink added)
Again, the ASPR has the authority to carry out drills and exercises, some which may be “without notice.” The National Health Security Strategy 2019-2022 has the sub-objective to “convene a unified, national response and recovery” while the ASPR Strategic Plan 2020-2023 has the plan to “fully implement, test, and validate the various components of the ASPR Incident Response Framework … including … ongoing exercising” of “operational doctrine” and “establish a … multi-year and multi-level exercise program designed to … achieve and sustain national preparedness to … respond to, and recover from all-hazards disasters,” which would include pandemics.Convene a Unified, National Response and Recovery to Public Health Emergencies and Disasters
ASPR, in coordination with HHS and Federal partners, leads the nation’s public health and medical preparedness and response and its health and social services recovery efforts … We will sustain efforts to organize, train, equip, and exercise response capabilities. We will identify health care readiness standards to assess coalition readiness status and ensure exercises test system resilience against routine and catastrophic threats. (Page 10; emphasis added)
Now, in the FBI’s own words, “The FBI is a primary law enforcement agency for the U.S. government.” And the aforementioned law which apparently allows hoaxes to be performed by such law enforcement agencies may be one reason why, in 2016, President Obama ordered the FBI to link “public health and law enforcement, and coordinate with INTERPOL [The International Criminal Police Organization] on the GHSA [Global Health Security Agenda] and its successful implementation.”any lawfully authorized investigative, protective, or intelligence activity of a law enforcement agency of the United States, a State, or political subdivision of a State, or of an intelligence agency of the United States (18 U.S. Code § 1038)
The emphasized part exempts activities that are not a violation of criminal laws. So politicians, public health officials, and other federal bureaus and departments may first attempt to ensure that their planned falsified pandemic exercise would not be a violation of criminal laws. Either way, it would still be an act of terrorism (deaths due to unnecessary use of ventilators and “vaccines,” closure of Churches, schools, and businesses, draconian lockdowns, coerced vaccinations, etc.) but the attempt at legalizing the act may prevent future legal action.the term “domestic terrorism” means activities that—
(A) involve acts dangerous to human life that are a violation of the criminal laws of the United States or of any State;
(B) appear to be intended—
(i) to intimidate or coerce a civilian population;
(ii) to influence the policy of a government by intimidation or coercion; […] (18 U.S. Code § 2331, emphasis added)


Can't get any bad reviews if you don't allow any reviews. Genius. pic.twitter.com/CsLi7ma3cM
— Paul Joseph Watson (@PrisonPlanet) October 11, 2021
Fauci audience score just dropped lmfao pic.twitter.com/CMNVrG2OrD
— Lauren Southern (@Lauren_Southern) October 11, 2021

#infowars shirt spotted! #FuckFauci pic.twitter.com/Qh9P2HloOf
— Racheal Rae (@LaehcaRae) October 11, 2021



Rajesh Gandhi is on the advisory board of Gilead Sciences.
David Glidden is a consultant for Gilead Sciences.
Adaora Adimora is a consultant for Gilead Sciences and received research support from Gilead Sciences.
Eric Daar is a consultant for Gilead Sciences and recieves research support from Gilead Sciences.
Judith Aberg received research support from Gilead Sciences.
Jason Baker received research support from Gilead Sciences.
Susanna Naggie received research support from Gilead Sciences.
Pablo Tebas received research support from Gilead Sciences.
Roger Bedimo received an honoraria from Gilead Sciences.
Please do not miss the fact that there were reported 71 percent adverse events in the 5-day study and 74 percent adverse events in the 10-day study for patients taking Veklury® (remdesivir). 21 percent suffered serious adverse events in the 5 day study and 35 percent of the patients suffered serious adverse events in the 10-day study. Below is the chart of adverse events published in the study.In a randomized, open-label clinical trial (Study GS-US-540-5773) of remdesivir in 397 subjects with severe COVID-19 treated with remdesivir for 5 (n=200) or 10 days (n=197), adverse events were reported in 71 percent and 74 percent of subjects, respectively, serious adverse events were reported in 21 percent and 35 percent of subjects, respectively, and Grade=3 adverse events were reported in 31 percent and 43 percent of subjects, respectively. Nine (5 percent) subjects in the 5-day group and 20 (10 percent) subjects in the 10-day group discontinued treatment due to an adverse event. All cause mortality at Day 28 was 10 percent vs 13 percent in the 5- and 10-day treatment groups, respectively.


Notice the differences in reporting. The May 2020 report describes adverse events, whereas the October 2020 report changes the reporting to adverse reactions.Study GS-US-540-5773 was a randomized, open-label clinical trial in hospitalized adult subjects with severe COVID-19 treated with VEKLURY 200 mg on Day 1 and 100 mg once daily for 5 (n=200) or 10 days (n=197). Adverse reactions were reported in 33 (17 percent) subjects in the 5-day group and 40 (20 percent) subjects in the 10-day group. The most common adverse reactions occurring in at least 5 percent of subjects in either the VEKLURY 5-day or 10-day group, respectively, were nausea (5 percent vs 3 percent), AST increased (3 percent vs 6 percent), and ALT increased (2 percent vs 7 percent). Rates of any adverse reaction, serious adverse reactions, and adverse reactions leading to treatment discontinuation are presented in Table 6. [Chart 6 indicated that 3 percent of the 5 day group and 5 percent of the 10 day group had treatment discontinued due to adverse reactions.]
This is where the deception becomes obvious. The study did not measure aderse reactions! The study protocols for GS-US-540-5773 published by Gilead states that they were only going to measure adverse events. There is no mention of any plan to measure advers reactions.May 2020 FDA Publication: “397 subjects with severe COVID-19 treated with remdesivir for 5 (n=200) or 10 days (n=197), adverse events were reported in 71 percent and 74 percent of subjects”
October 2020 FDA Publication: “Adverse reactions were reported in 33 (17 percent) subjects in the 5-day group and 40 (20 percent) subjects in the 10-day group.”
May 2020 FDA Publication: Nine (5 percent) subjects in the 5-day group and 20 (10 percent) subjects in the 10-day group discontinued treatment due to an adverse event.
October 2020 FDA Publication: 3 percent of the 5 day group and 5 percent of the 10 day group discontinued treatment due to an adverse reaction.
May 2020 FDA Publication: Serious adverse events were reported in 21 percent and 35 percent of subjects, [in the 5 day and 10 day groups] respectively.
October 2020 FDA Publication: Serious adverse reactions were reported in 2 percent and 2 percent of subjects in the 5 day and 10 groups respectively.







